Infusion Pump Software Issue from Baxter
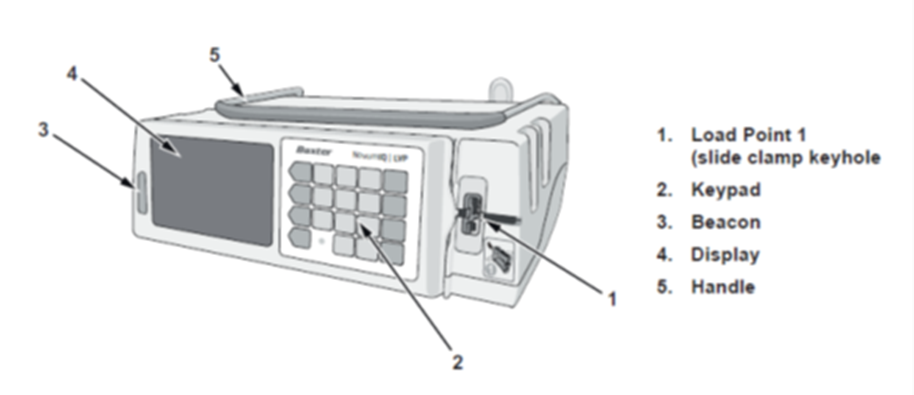
Affected Product
The FDA is aware that Baxter has issued a letter to affected customers recommending all Novum IQ Large Volume Pumps (LVP) and Novum IQ Syringe Pumps (SP) be corrected prior to continued use.

Device Use
The Baxter Novum IQ LVP and Novum IQ Syringe Pump are intended to provide intravenous infusion of parenteral fluids, blood, and blood products to a patient under the direction or supervision of a physician or other certified healthcare professional.
What to Do
While providing patient care involving high-risk medications and/or critical illness, users should consider utilizing an alternate pump, if available and appropriate for the patient’s treatment. Customers can continue to use the Novum IQ LVP and Novum IQ Syringe Pump while following the instructions found here.
