FDA MedWatch Early Alert: Dialysis Tubing Set Issue from Vantive
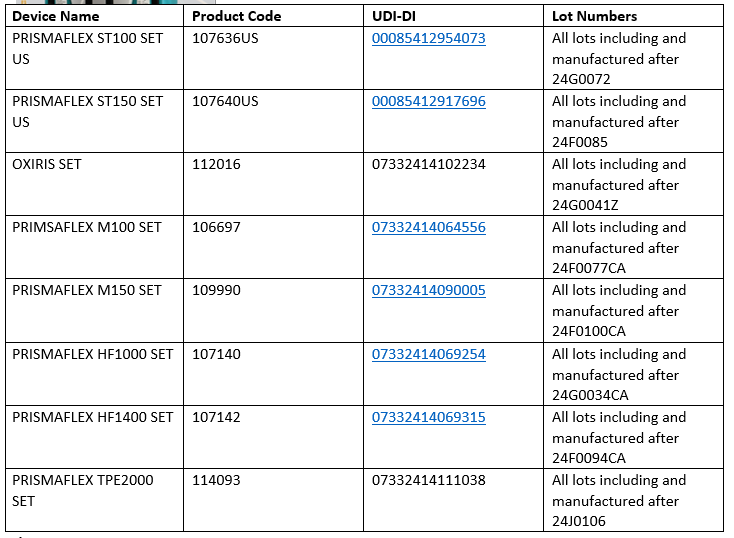
Affected ProductThe FDA is aware that Vantive has issued a letter informing affected customers that Prismaflex Control Units have updated use instructions. |
 |
Reason for AlertVantive stated that Prismaflex Set deaeration chambers may dislodge from the Prismaflex Control Unit. This issue does not affect PrisMax Control Units, as these devices have a different deaeration chamber holder design. A dislodged deaeration chamber may lead to an “Air in Blood” alarm on the Prismaflex Control Unit. The alarm may occur during the priming phase prior to initiating therapy, or during therapy. This may result in delays or interruptions to therapy and blood loss due to clotting and failure to return blood manually. As of December 17, Vantive has not reported any serious injuries or deaths associated with this issue. The FDA is currently reviewing information about this potentially high-risk device issue and will keep the public informed as significant new information becomes available. |
Affected Devices |